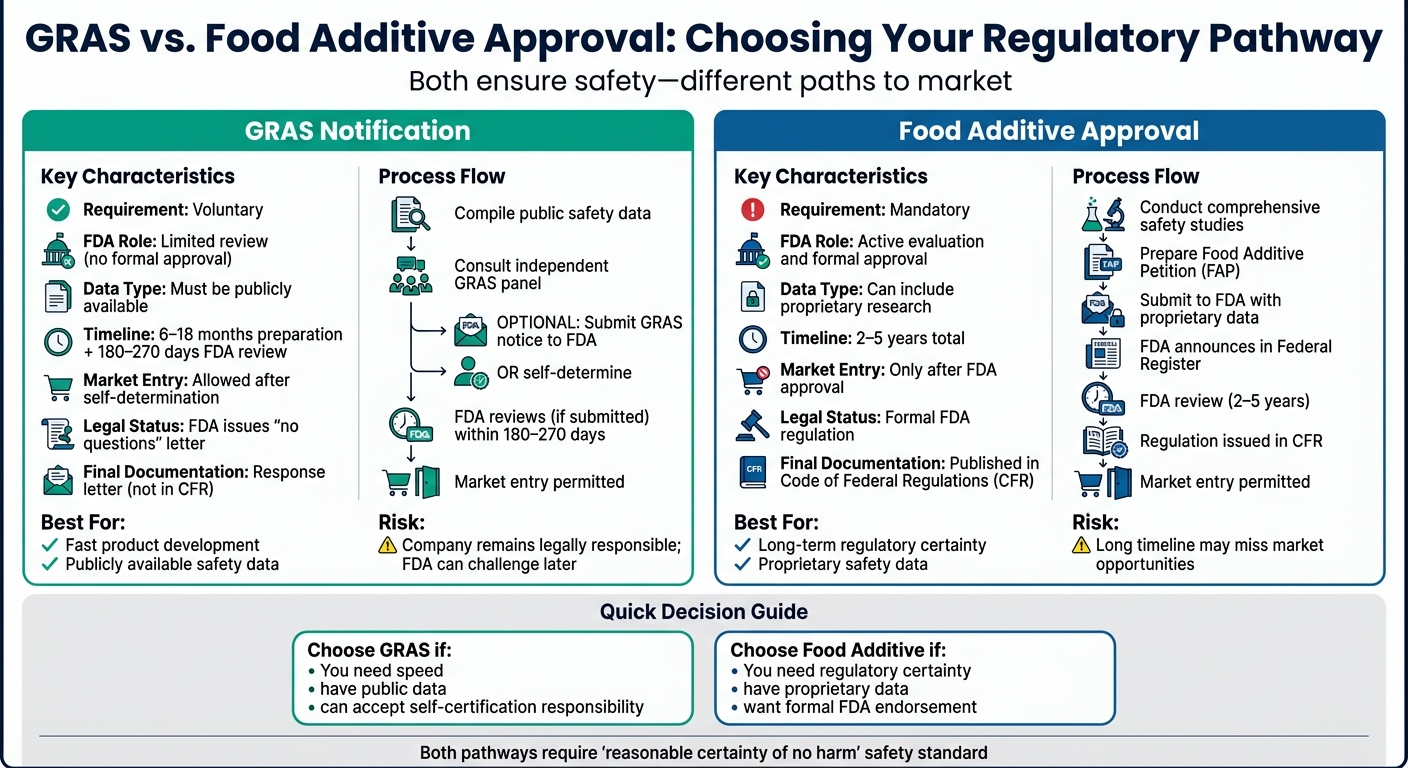
Every food ingredient in the U.S. must meet strict FDA safety standards. Manufacturers and students in culinary courses can choose between two regulatory pathways: GRAS (Generally Recognized as Safe) or Food Additive Approval. Both ensure safety but differ in process, cost, and FDA involvement.
- GRAS: Faster, voluntary, relies on public data, and allows market entry after self-determination. FDA may review but doesn’t formally approve.
- Food Additive Approval: Mandatory, slower (2–5 years), requires proprietary data, and results in formal FDA regulation in the Code of Federal Regulations (CFR).
Quick Comparison
| Feature | GRAS Notification | Food Additive Approval |
|---|---|---|
| Requirement | Voluntary | Mandatory |
| FDA Role | Limited review | Active evaluation |
| Data Type | Publicly available | Can include proprietary |
| Timeline | ~6–18 months (+180–270 days FDA review) | 2–5 years |
| Market Entry | Allowed after self-determination | Only after FDA approval |
Key takeaway: GRAS is faster but carries more risk, while Food Additive Approval offers long-term regulatory certainty.
GRAS vs Food Additive Approval Process Comparison Chart
What Is the GRAS Notification Process?
What GRAS Means
GRAS stands for "Generally Recognized as Safe" [1]. Under the FD&C Act, substances with GRAS status are not classified as food additives, which means they bypass the mandatory pre-market approval process required for additives [1].
Everyday ingredients like canola oil, vinegar, and spices such as black pepper are examples of GRAS substances as of June 2024 [1]. Their safety is either backed by a long history of use in food or by publicly available scientific research. The main condition is that qualified experts must generally agree the substance is safe, based on accessible data.
"For the use of an ingredient to be considered GRAS, all data necessary to establish safety must be publicly available and its safe use must be generally recognized by qualified experts." – FDA [1]
How the GRAS Notification Process Works
The GRAS designation allows for a simplified notification process. While voluntary, the FDA encourages companies to participate [1]. To begin, companies collect safety data on the substance, including its identity, production methods, intended technical effects, and estimated consumption levels. Often, manufacturers consult an independent GRAS panel to evaluate the evidence.
After compiling the data, companies have two options: they can submit a GRAS notice to the FDA or make an independent determination (commonly called "self-determined GRAS"). Companies opting for self-determined GRAS can market their products without notifying the FDA but remain legally responsible for ensuring their ingredients are safe and compliant with regulations [1].
What the FDA Does in the GRAS Process
The FDA plays a unique role in the GRAS process, distinct from its responsibilities in food additive approvals. When a company submits a voluntary GRAS notice, the FDA reviews it to confirm whether the provided evidence supports the safety conclusion. However, the agency does not "approve" GRAS substances [1]. Instead, it issues a response letter indicating either that the FDA has "no questions" about the GRAS determination or that the notice lacks sufficient evidence to support the claim.
The FDA aims to respond within 180 days, though it may extend this timeline by an additional 90 days [2]. In reality, the process has sometimes taken up to a year for final responses [3]. For transparency, all GRAS submissions and the FDA’s response letters are made publicly available on the agency’s website, ensuring openness in the food supply chain.
sbb-itb-7261261
What Is the Food Additive Approval Process?
What Food Additive Approval Means
Getting approval for a food additive isn’t optional – it’s the law. According to the FDCA, any food substance that isn’t considered Generally Recognized as Safe (GRAS) must receive pre-market authorization from the FDA before it can be sold [1][5]. This requirement was introduced by the 1958 Food Additives Amendment, which shifted responsibility to manufacturers. They must prove with evidence that a substance poses "reasonable certainty of no harm" before it enters the food supply [1][6].
Using an unapproved ingredient is a serious violation. Products containing such ingredients are labeled adulterated and can’t legally be sold in the U.S. Tim Lombardo, Senior Director of Food Consulting Services at EAS Consulting Group, explains:
"Any unapproved food ingredients used in a food or beverage render the product adulterated under the Food, Drug & Cosmetic Act, making it illegal to import or market in the United States" [4].
How the Approval Process Works
The approval process starts with the manufacturer submitting a Food Additive Petition (FAP) to the FDA. This petition must include a range of detailed information, such as:
- The chemical makeup and production methods of the substance
- How the additive will be used and its intended effects
- Estimated consumption levels across different demographics
- Comprehensive safety studies, including laboratory, animal, and sometimes human research
- An Environmental Impact Statement to evaluate the potential effects of production and waste [4][6]
Most of the safety data in these petitions comes from unpublished and proprietary research conducted or sponsored by the company [6].
Once the petition is submitted, the FDA announces it in the Federal Register. For additives intended for meat or poultry products, the agency also consults with the U.S. Department of Agriculture [1].
If the FDA approves the petition, it issues a regulation in the U.S. Code of Federal Regulations (CFR). This regulation outlines how the additive can be used, including the types of food it can be added to, the maximum allowable amounts, and any labeling requirements [4][1]. For instance, in June 2024, the FDA approved xanthan gum for use as a texturizer in products like salad dressings, chocolate milk, bakery fillings, and puddings, with clear labeling rules [1].
What the FDA Does in the Approval Process
The FDA plays a hands-on role in the food additive approval process, unlike the more passive approach it takes with GRAS substances. FDA scientists rigorously review all the submitted data to ensure the additive meets the safety standard of "reasonable certainty of no harm" [1]. This evaluation includes examining how the body processes the substance – covering absorption, digestion, and excretion – and determining an Acceptable Daily Intake (ADI). The ADI includes a safety margin to protect vulnerable groups like children and pregnant individuals [1].
Legally, the FDA has 180 days to respond to a petition. However, follow-up questions and requests for additional data often extend the timeline [6]. In reality, the process typically takes 2 to 5 years from the initial submission to the final decision [4]. If the FDA concludes that an additive fails to meet safety standards, it can issue warning letters, alert the public, or take enforcement action to halt the distribution of the ingredient and any products containing it [1].
Main Differences Between GRAS and Food Additive Approval
Safety Standards Compared
Both GRAS (Generally Recognized as Safe) and food additive approval processes hinge on the same safety benchmark: "reasonable certainty of no harm" for their intended use [1]. The FDA emphasizes that GRAS substances must meet the same rigorous safety standards as food additives, requiring equivalent levels of evidence and data quality [1]. However, the key distinction lies in how the safety data is evaluated and accessed. For GRAS status, the evidence must be publicly available and widely acknowledged by qualified experts. On the other hand, food additive petitions can include proprietary or confidential research submitted directly to the FDA for review [1]. These differences shape the level of FDA involvement in each pathway.
FDA Oversight Compared
The FDA plays a more hands-on role in food additive approval compared to the GRAS process. For food additives, manufacturers are legally required to secure FDA approval before marketing their ingredient [1]. This involves submitting a detailed petition, which the FDA reviews thoroughly before issuing a formal regulation in the Code of Federal Regulations (CFR), outlining how the additive can be used [1].
The GRAS process, in contrast, is voluntary. Manufacturers can independently determine that a substance is GRAS and begin using it in food products without notifying the FDA [1][2]. Christopher M. Lahiff, Alcohol Law Advisor at McDermott Will & Schulte LLP, explains:
"Manufacturers remain free to conduct their own GRAS evaluations and then proceed to incorporate the substance into food" [2].
If a manufacturer does choose to submit a GRAS notification, the FDA’s role is limited to reviewing the conclusion and issuing a response letter – typically stating whether the agency has any questions [1].
| Feature | Food Additive Approval | GRAS Notification |
|---|---|---|
| Legal Requirement | Mandatory pre-market approval [1] | Voluntary notification [1][2] |
| FDA Involvement | Active evaluation and approval [1] | Limited review of manufacturer’s conclusion [1] |
| Evidence Basis | FDA review of submitted petition [1] | General recognition by qualified experts [1] |
| Information Status | Can include proprietary data [1] | Must be publicly available [1] |
| Final Authorization | Regulation issued in the CFR [1] | FDA response letter (e.g., "no questions") [1] |
Flexibility and Cost Compared
The GRAS pathway offers manufacturers a faster and more flexible route to market. Since pre-market approval isn’t required, companies can self-determine GRAS status and launch products more quickly – a significant advantage in competitive industries with tight development timelines [2]. Food additive approval, however, is a slower and more expensive process. But it comes with the benefit of formal FDA endorsement, which provides a clear legal framework. This includes regulations in the CFR that may specify usage limits, applicable food types, and labeling requirements [1]. Such regulatory clarity can reduce legal risks and enhance consumer confidence.
That said, manufacturers who skip the voluntary GRAS notification process take on full responsibility for ensuring their products meet safety standards. If a self-affirmed GRAS substance is later found to pose safety risks, the FDA can step in with enforcement actions [1].
GRAS Made Simple: Navigating FDA Ingredient Safety
Timeline Differences
The difference in timelines between GRAS notifications and food additive approvals plays a significant role in how quickly products can hit the market. Understanding these variations is crucial when deciding on the right regulatory pathway.
How Long GRAS Notifications Take
The GRAS assessment process generally takes 6 to 18 months before a notice is submitted to the FDA [4]. This phase includes compiling safety evidence, convening an independent expert panel, and reaching a consensus on the substance’s safety for its intended use.
Once the GRAS notice is filed, the FDA typically responds within 180 days, though this period can be extended by an additional 90 days if needed [7]. Attorney Riëtte van Laack highlights this timeline:
"FDA commits to respond to the notifier within 180 days of the date of filing the GRN or AGRN, with a possible extension by another 90 days." [7]
One key advantage of the GRAS route is the "Self-Determined GRAS" option. If an independent expert panel concludes that the substance is safe, manufacturers can begin marketing the ingredient in the U.S. immediately, even without notifying the FDA. While notification is voluntary, it provides added regulatory assurance [4].
On the other hand, food additive petitions involve a much longer process.
How Long Food Additive Approvals Take
Food additive petitions usually require 2 to 5 years from submission to final approval [4].
Tim Lombardo, Senior Director of Food Consulting Services at EAS Consulting Group, explains:
"Obtaining a GRAS certification or completing a Food Additive Petition is complex; GRAS generally takes 6-18 months, while FAP typically takes 2-5 years." [4]
This extended timeline is due to the formal notice-and-comment rulemaking process. Unlike GRAS notifications, food additives cannot be marketed until the FDA issues a final regulation in the Code of Federal Regulations [1]. For manufacturers with tight timelines or competitive launches, this delay can be a significant hurdle.
Here’s a quick comparison of the timelines:
| Feature | GRAS Notification | Food Additive Petition (FAP) |
|---|---|---|
| Preparation Timeline | 6–18 months for assessment and expert review [4] | Requires original lab, animal, and human studies [4] |
| FDA Review Timeline | 180–270 days [7] | 2–5 years [4][7] |
| Market Entry | Allowed after self-determination by experts [4] | Prohibited until FDA publishes a final regulation [1] |
| FDA Outcome | "No questions" letter (not a formal approval) [4] | Formal regulation/authorization in the CFR [1] |
How These Processes Affect the Food Industry
The decision between GRAS notification and food additive approval shapes how quickly companies can innovate, manage compliance costs, and handle risks over time. By understanding the differences in their requirements and timelines, it becomes clear how each option influences product development and market strategies.
Why GRAS Works Well for Fast Product Development
The GRAS pathway is a go-to option for companies aiming to speed up product launches. Since pre-market approval isn’t needed, businesses can introduce products as soon as they confirm a substance’s safety [1]. This is a game-changer for startups and smaller players trying to stay competitive in fast-moving markets.
Christopher M. Lahiff, Alcohol Law Advisor at McDermott Will & Schulte LLP, highlights this advantage:
"If an ingredient is GRAS, food additive petition is not required and FDA does not have to approve the ingredient before it can be used in foods." [2]
On the flip side, the food additive approval process prioritizes regulatory predictability, though it often comes at the expense of speed.
Why Food Additive Approval Provides More Certainty
While GRAS is all about speed, the food additive approval route offers a clear legal framework. With this process, the FDA provides formal endorsement by issuing a regulation in the Code of Federal Regulations (CFR). This regulation specifies exactly how the additive can be used, including permissible food types, usage levels, and labeling requirements [1].
For companies focused on long-term planning or operating in highly regulated markets, this certainty is invaluable. It minimizes the risk of future regulatory disputes, giving businesses more confidence in their ingredient choices.
Risks to Consider
Both pathways come with risks, and companies need to manage them carefully. For GRAS, the main concern is post-market enforcement. Even if a company determines an ingredient is GRAS, the FDA can later challenge that conclusion. If the agency finds safety standards unmet or public recognition lacking, the substance is treated as an unapproved food additive. This could lead to warnings or even halting product distribution [1].
The FDA emphasizes this point:
"Manufacturers that choose not to go through the FDA’s GRAS Notification program are still responsible to produce products that are compliant with the law." [1]
For food additives, the lengthy approval process poses a different kind of risk. With approvals often taking 2 to 5 years, market conditions could shift dramatically, potentially undermining the business case for the ingredient by the time it’s approved [1].
Choosing the Right Pathway
When deciding between regulatory pathways, it’s essential to weigh factors like data availability, timeline priorities, and the level of regulatory certainty you need. Your choice ultimately depends on how you plan to demonstrate safety and what you require from the FDA.
Start by evaluating your data. If your safety data is proprietary, the food additive route is your only option [1]. GRAS status demands publicly accessible safety data, which means companies relying on trade secrets or unpublished research can’t take this route. This distinction is crucial because the type of data you possess directly impacts both the process you can pursue and the regulatory outcome you’ll achieve.
If time is a concern, GRAS offers a faster option. The FDA typically responds to GRAS notifications within 180 to 270 days, making it a quicker process [2]. In contrast, food additive approvals involve formal rulemaking, complete with Federal Register notices, which can significantly extend the timeline. While GRAS provides an FDA response letter, food additive approval results in a formal regulation in the Code of Federal Regulations (CFR), specifying exactly how the ingredient can be used [1]. This speed-versus-certainty trade-off is a key factor in your decision.
For long-term clarity, food additive approval offers more regulatory certainty. When the FDA publishes a food additive regulation in the CFR, it establishes clear guidelines, such as permitted uses, maximum levels, and labeling requirements. This creates a solid legal framework that can protect against future regulatory challenges. On the other hand, GRAS status provides flexibility and faster market entry but leaves companies responsible for defending their safety conclusions if the FDA revisits them later [1].
Additionally, check if your ingredient qualifies as a "prior-sanctioned substance" – those approved before 1958 are exempt from the food additive definition [1]. Keep in mind that modifying an approved food additive’s use or level requires submitting a new petition [1]. Your decision should balance immediate market needs with the desire for long-term regulatory stability.
FAQs
When should I choose GRAS instead of a food additive petition?
When deciding between regulatory pathways for food ingredients, GRAS (Generally Recognized As Safe) offers a quicker and less formal option. If your ingredient is already considered safe by qualified experts for its intended use, GRAS allows you to independently confirm its safety by relying on existing scientific data. This process doesn’t require prior FDA approval, streamlining the timeline.
On the other hand, submitting a food additive petition is a more rigorous route. It involves compiling and submitting extensive data for FDA review, which can be both time-intensive and complex.
What makes a GRAS determination legally defensible?
A GRAS (Generally Recognized as Safe) determination holds up legally when it’s backed by a detailed scientific safety evaluation conducted by qualified experts. This evaluation must demonstrate a reasonable certainty of no harm for the ingredient’s intended use. Key components of this process include toxicology data, dietary exposure analysis, and metabolic studies. A clear and well-documented review – whether carried out by independent experts or through FDA notification – ensures adherence to regulatory requirements and establishes a solid, defensible record.
What happens if FDA later challenges a self-determined GRAS ingredient?
If the FDA disputes a self-determined GRAS (Generally Recognized as Safe) ingredient, it has the authority to take regulatory action. This might involve mandating that the ingredient undergo the formal food additive approval process or even pulling it from the market entirely. Additionally, the FDA’s findings could lead to revisiting or potentially revoking the self-affirmation pathway for that ingredient.